Strain improvement for increasing Cellobiohydrolase I (CBH1) production in S. cerevisiae
When it comes to strain improvement for achieving the best protein yields, simply increasing the copy number or strength of the target protein expression cassette often does not cut it. Strain engineering requires a comprehensive approach that combines improving the target protein expression along with optimization of the genetic background of the host. The traditional methods of strain improvement – such as classical mutagenesis – can get you to the end goal but run up long development times and carry the risk of accumulating mutational load with potentially unpredictable downstream effects.
Cellobiohydrolase I (CBH1), an enzyme involved in the degradation of lignocellulose is being actively explored for consolidated bioprocessing applications. In this experiment, we expressed the codon-optimized fungal CBH1 gene is S. cerevisiae with the corresponding native signal sequence under the control of a yeast ENO2 promoter. Then, we designed several concept libraries spanning diverse targets and edit types, for a total of 14,859 edits across the genome.
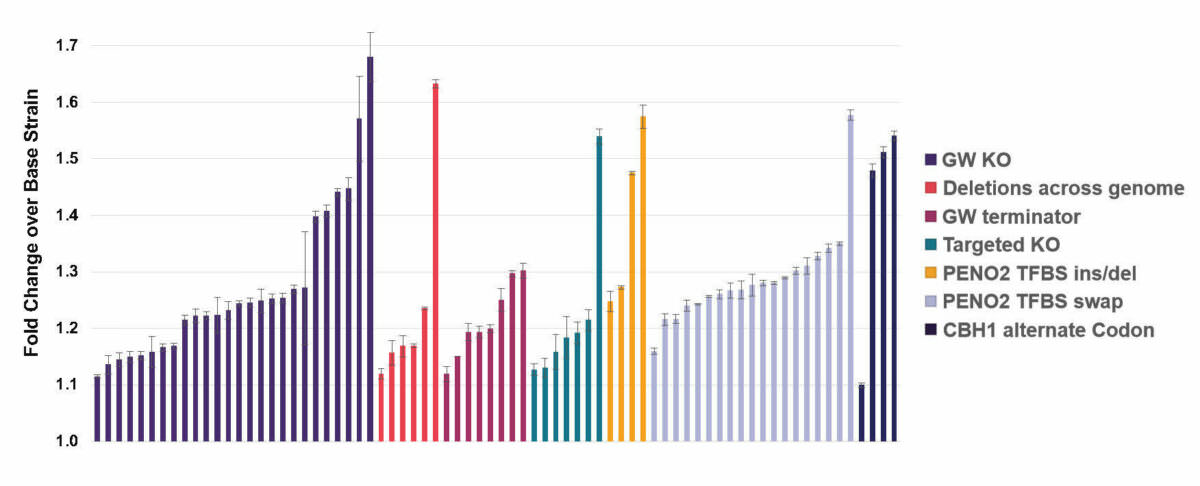
Activity improvement in edited strains across different libraries.
After screening several thousands of isolates from different libraries, we identified 74 unique hits that improved production of CBH1 in our strain. These included previously known and novel targets across a wide range of functions: protein degradation and secretion, stress response, transcription and translation, nuclear transport, and uncharacterized genes. These hits came from both rational and exploratory library types, including transcription factor binding site and terminator engineering, genome-wide knockouts and short deletions, targeted knockouts, and alternate codon libraries.
Generate any library at the push of a button.
The Onyx gives you the flexibility and scalability to develop virtually limitless libraries. Create insertions, deletions, substitutions, or full saturation mutagenesis, in a single gene, a pathway, or the whole genome.
- Copyright © 2025 Inscripta, Inc. All rights reserved.
- Legal Notices
- Privacy Policy
