Identifying genome-wide targets of osmotic stress tolerance in E. coli using CRISPR-mediated forward engineering
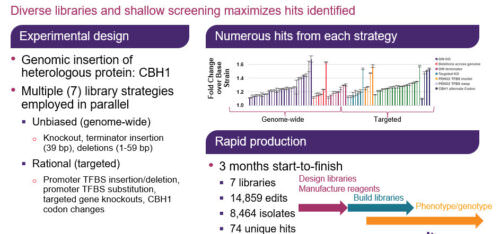
Biological responses to environmental stress can arise from multiple genetic solutions encoding tolerance mechanisms. In order to investigate the response to osmotic pressure in E. coli, we used an automated CRISPR editing workflow to generate a diverse cell population that included gene knockouts and different strength promoter substitution libraries targeting almost every gene in the genome. The resulting 25,000-variant pooled library was studied under a titrated salt challenge and the population dynamics were tracked using plasmid barcodes. By mapping 300,000 data points, we identified clear enrichment and depletion patterns and connected those back to functional genome annotations. We were able to rapidly validate our experiment with the performance of known salt stress tolerance loci, such as rpoE and osmE, and discover new gene targets.