High-throughput CRISPR editing using the Onyx platform identifies essential residues in proteins
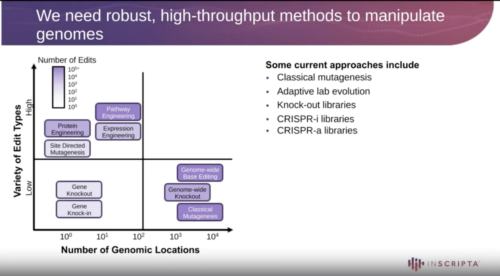
In this study, we used the high-throughput CRISPR-based Onyx platform to perform saturation mutagenesis on four different essential genes involved in cell envelope synthesis in E. coli. 22,790 edits were designed. We used these saturation mutagenesis libraries to probe the essentiality of all different residues. Edits that could not be introduced suggests those residues are essential for protein function. We identified known essential amino acids (i.e. catalytic residues and residues involved in substrate binding), thereby validating our experimental approach. Several residues that were previously not known to be essential were identified. We expect our results to offer vastly improved insights into protein function, help fight against antibiotic resistance and aid structure-based drug design targeted against these essential proteins.